 Weinhold and Landis describe the symmetry adapted use of the orbital hybridization concept within the context of natural bond orbitals, a localized orbital theory containing modernized analogs of classical (valence bond/Lewis structure) bonding pairs and lone pairs. As a result of later developments resulting partially from the introduction of VSEPR, an alternative view arose which considers the two lone pairs to be equivalent, colloquially called rabbit ears. Initially, Linus Pauling's scheme of water as presented in his hallmark paper on valence bond theory consists of two inequivalent lone pairs of σ and π symmetry. Multiple bonds įurther information: Lone pair The symmetry-adapted and hybridized lone pairs of H 2O Nevertheless, as pointed out in Carroll's textbook, at lower levels of theory, the two models make different quantitative and qualitative predictions, and there has been considerable debate as to which model is most useful conceptually and pedagogically. Ian Fleming goes further in a 2010 textbook, noting that "the overall distribution of electrons is exactly the same" in the two models. Wiberg concluded that "although a conclusive statement cannot be made on the basis of the currently available information, it seems likely that we can continue to consider the σ/π and bent-bond descriptions of ethylene to be equivalent. Two equivalent orbitals h and h' can be constructed by taking linear combinations h = c 1σ + c 2π and h' = c 1σ – c 2π for an appropriate choice of coefficients c 1 and c 2. The two representations produce the same total electron density and are related by a unitary transformation of the occupied molecular orbitals different localization procedures yield either of the two. The σ-π treatment takes into account molecular symmetry and is better suited to interpretation of aromatic molecules ( Hückel's rule), although computational calculations of certain molecules tend to optimize better under the equivalent-orbital treatment. 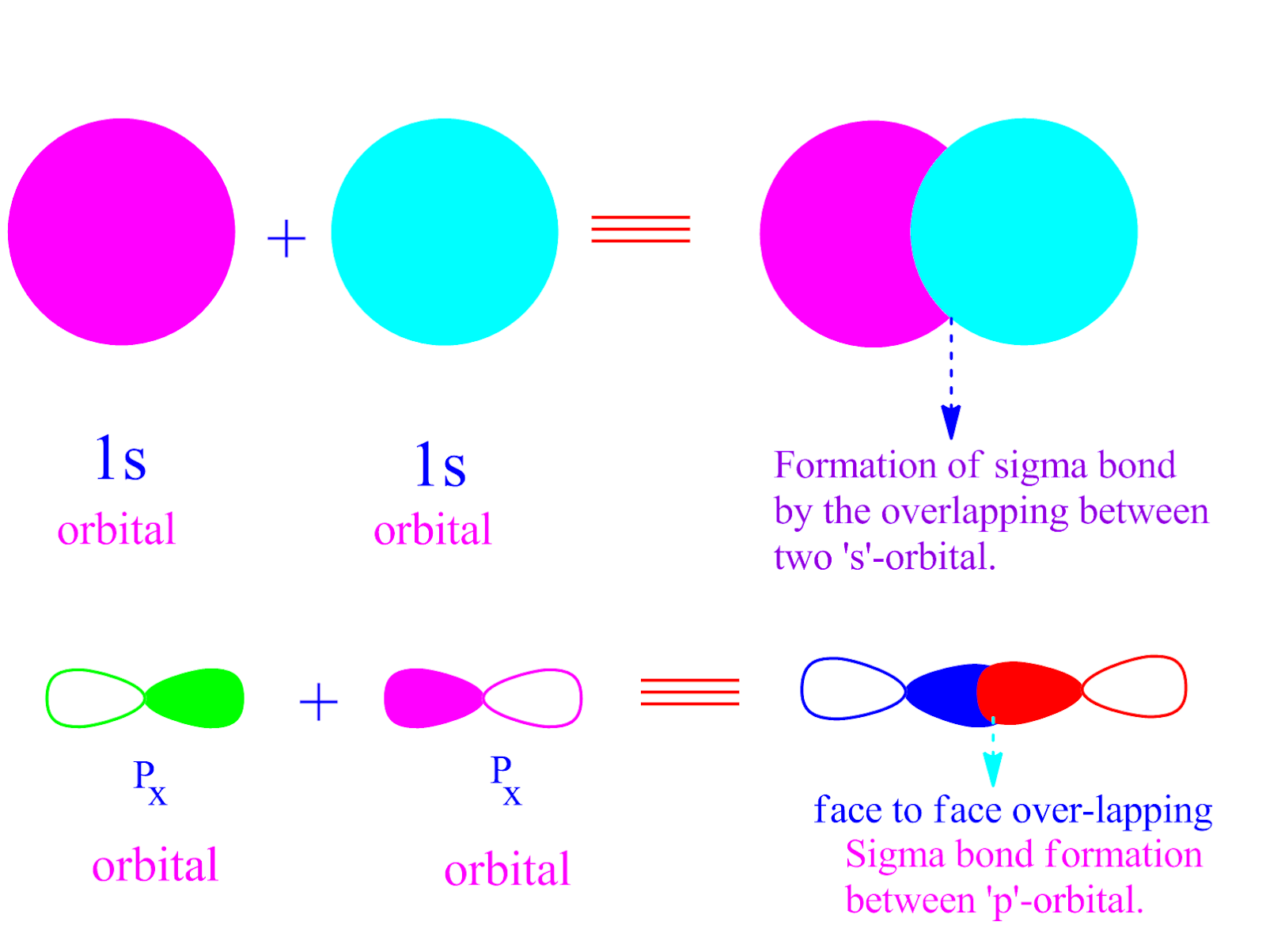 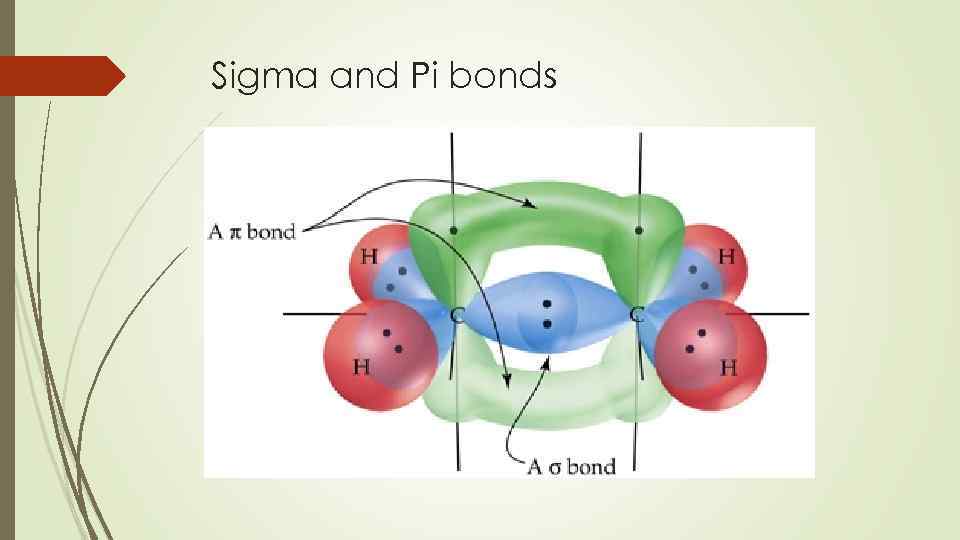 
The σ-π model differentiates bonds and lone pairs of σ symmetry from those of π symmetry, while the equivalent-orbital model hybridizes them. The σ-π model and equivalent-orbital model refer to two possible representations of molecules in valence bond theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
